So what happens in your body when you take cannabis or cannabinoids?
This primarily depends on which cannabinoids were taken and how they were taken/the route of administration.
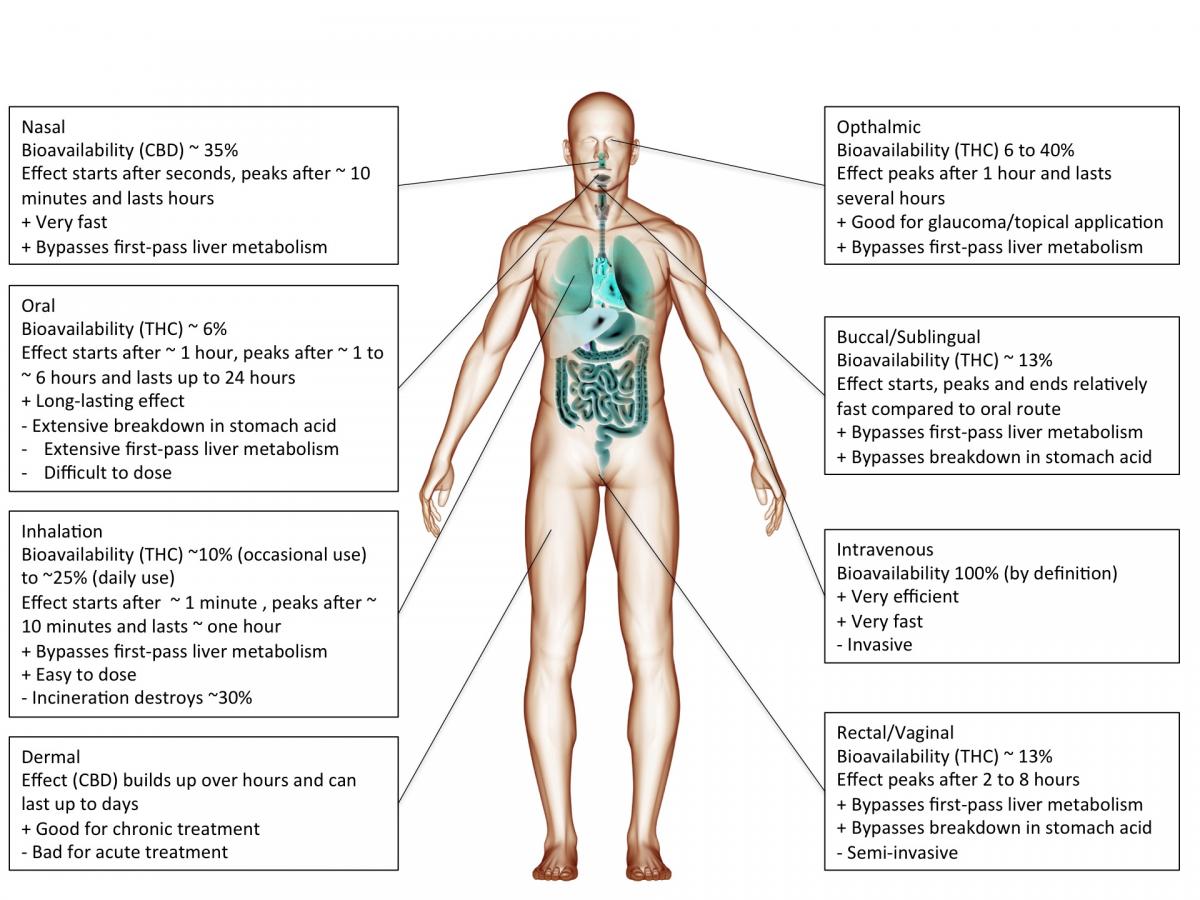
Cannabis or cannabinoids are often smoked but can also be ingested (oil, cake, tea etc.), injected or absorbed through the skin (patch) or mucous membranes (tongue, cheek, rectum). The chosen route of administration has a major impact on the actual effect.
For instance, smoking inevitably heats up cannabinoids causing decarboxylation and a shift from acidic to neutral compounds, which tend to be more biologically active. In addition, the incineration process will render part of the cannabinoids biologically inactive.
However, inhaled cannabinoids enter the bloodstream through the lungs and therefore reach the rest of the body before it passes the liver. This first-pass liver effect can seriously reduce the amount of available cannabinoids. Similarly, ingesting cannabinoids can reduce the amount of available cannabinoids through degradation in the gut/digestive tract.
Heat, chemical degradation or biological degradation/metabolism can dramatically change the amount of cannabinoids that is biologically active: bioavailability. Injected substances are considered 100% bioavailable. Any reduction is ascribed to the route of administration.
Smoked, or inhaled cannabinoids have reported bioavailabilities ranging from 2-56%(Huestis, 2007) with an average of about 30% (McGilveray, 2005). This variability is mainly due to differences in smoking dynamics (how deeply does one inhale, how long does one hold it in). Compared to oral application, the effects of smoking cannabinoids are relatively fast in on- and offset.
cannabinoids that are absorbed through the mucous membranes in the mouth (buccomucosal application) have bioavailabilities of around 13% (Karschner et al., 2011). Application via the mucous membranes of tongue and cheek or rectal application bypasses degradation in the gut and improves bioavailability.
When cannabinoids are ingested, bioavailability is reduced to about 6% (Karschner et al., 2011).
Literature
Huestis, M.A. (2007). Human cannabinoid Pharmacokinetics. Chem. Biodivers. 4, 1770–1804.
Karschner, E.L., Darwin, W.D., Goodwin, R.S., Wright, S., and Huestis, M.A. (2011). Plasma cannabinoid pharmacokinetics following controlled oral delta9-tetrahydrocannabinol and oromucosal cannabis extract administration. Clin. Chem. 57, 66–75.
McGilveray, I.J. (2005). Pharmacokinetics of cannabinoids. pain Res. Manag. J. Can. pain Soc. J. Société Can. Pour Trait. Douleur 10 Suppl A, 15A – 22A.